How will liquid biopsy change cancer diagnosis?
Liquid biopsy: a powerful new approach to cancer treatment, with huge market opportunities
Cancer has become a new focus of increasing concern in modern society. Various factors such as the ageing of the population, lifestyle, and the environment are driving the high incidence of cancer. According to the World Health Organization, one-sixth of the world's population die from cancer, and about one-half of people cause a variety of cancers throughout their life cycle. Cancer cells are genetically mutated cells; in order to study the mechanism of each cancer and determine the most appropriate method of diagnosis and treatment, these cancer cells need to be analyzed.
According to the consulting of McMasses, the traditional biopsy method requires invasive surgery to obtain some tissue of the tumor. However, these methods are costly and very painful for the patients, and therefore cannot perform regular routine operations to monitor the development of the tumor and improve long-term diagnosis and treatment. The emergence of "liquid biopsy" technology now allows us to abandon past invasive analysis methods.
In fact, only a small amount of blood from the patient needs to be collected, and relevant biomarkers can provide useful information about the patient's cancer status. Many companies are developing technologies to collect, isolate, and analyze circulating tumor DNA (ctDNA) and circulating tumor cells (CTCs) that diffuse into the blood system.
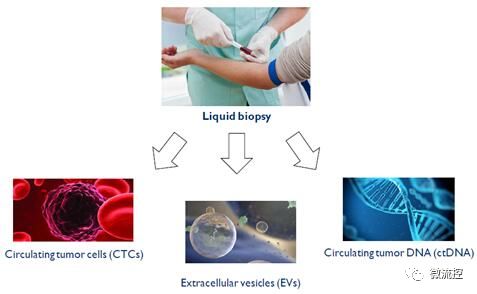
Liquid biopsy can analyze circulating tumor cells, circulating tumor DNA, and extracellular vesicles
Thanks to these new methods, not only cancers can be diagnosed non-invasively, but also the molecular mechanism of each patient, the selection and development of customized treatment methods, the monitoring of therapeutic effects (and drug resistance), and the recurrence of disease. Screening. Some companies are even studying early screening for cancer, although this is a longer-term type of application.
Therefore, liquid biopsy has the potential to be applied at all stages of cancer treatment, save more lives, and reduce medical costs. Its market potential is huge and the related market is already very large and is growing rapidly. According to the contents of this research report, the liquid biopsy market will grow from US$1.7 billion in 2017 to US$7.8 billion in 2023, and the compound annual growth rate during the period from 2017 to 2023 will reach 29%. This includes sales of hardware (systems and equipment) and reagents, as well as related service revenue for CTC and ctDNA applications.
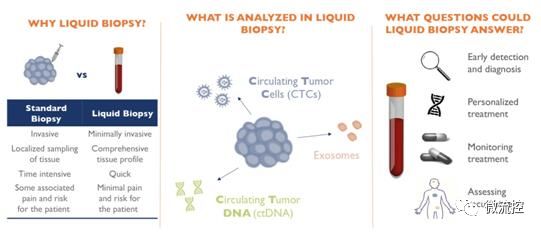
Liquid biopsy to change cancer treatment rules
Contested vendors and business models
It is not easy to study the variation of DNA in tumor cells. Many different equipment and techniques are needed. From the enrichment and analysis of biomarkers to the bioinformatics analysis of mutational spectroscopy, this industry chain is long and involves many vendors. This report clarifies all manufacturers at all levels of the industry chain, clarifies the best in each sector, and details their market value points, and how the development of liquid biopsy technology detonates the downstream analysis equipment market, such as sequencing and digital PCR and so on. This report also introduced the differences and complementarities between ctDNA and CTC. Yole analysts explained why the CTC has not yet begun to attract a lot of investment. In addition, thanks to the recent technological development, how will this situation change?
In fact, ctDNA vendors have attracted more than $3.7 billion in investment since 2010, while CTC companies have only raised $280 million. In terms of revenue, ctDNA vendors also have a significant advantage. This is mainly due to differences in business models: Most ctDNA vendors use their own methods developed in their own CLIA (United States Clinical Laboratory Improvement Amendment) laboratories without the approval of the FDA (US Food and Drug Administration). In addition, another advantage of ctDNA is that DNA is easier to store and transport than living cells. In contrast, most CTC vendors develop instrumentation and consumables that are sold directly to end users.
However, most of the instruments used in these CTC separation applications are still only used in the research phase because the FDA still needs more evidence from the patient's survival rate to justify the benefits of CTC analysis before approving their clinical application. This explains, at least in part, why CTC vendors lag behind ctDNA vendors in terms of revenue. Fortunately, thanks to recent technological developments, and more and more literature has proven the value of CTC analysis (in fact, intact living cells carry more information than DNA sequences), this situation will be very It will change soon.
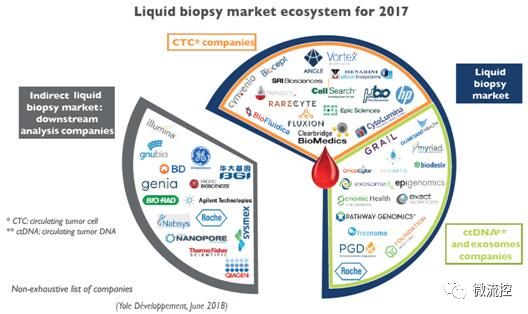
2017 Liquid Biopsy Market Ecosystem
Microfluidic technology promotes technological breakthrough
Thanks to the development of microfluidic technology, several steps in the liquid biopsy workflow have been realized. From separation to downstream analysis, microfluidic devices are ubiquitous. An important part of CTC enrichment and separation systems relies on microfluidic devices that separate CTCs from other blood cells. These microfluidic devices take advantage of the physical properties of the cell, such as size, shape, deformability, and dielectric properties.
Key manufacturers such as Angle plc, Celsee, Clearbridge Biomedics and Vortex Biosciences rely on microfluidic technology. These microfluidic protocols can be used for a wide range of cells for low-cost, high-throughput capture. In addition, downstream analysis solutions also need to rely on microfluidic technology in many cases: the DNA sequencers of companies such as Illumina and Ion Torrent use microfluidics to flow cells, others such as Bio-Rad, Stilla Technologies and RainDance ( Technologies such as Drop Digital PCR, acquired by Bio-Rad, also use microfluidics.
All of these state-of-the-art technologies need to rely on innovative microfluidic hardware to enable more sophisticated analysis functions, resulting in practical improvements in cancer care. In this report, Yole focuses on microfluidic technology at various stages of the liquid biopsy workflow.
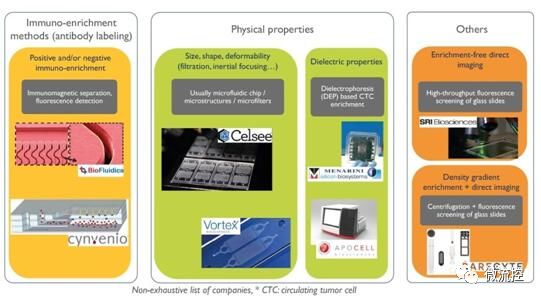
Liquid Biopsy Market: CTC Workflow Technology
200*300mm Anti-Blue Light Film
The Imported Anti-Blue Light For ipad is made of high-quality imported TPU material. It is precisely cut by the Screen Protector Cutting Machine. Protect your device screen from scratches and cracks. The flexible TPU Screen Protector can completely cover the iPad screen, providing the maximum protection for the iPad.
Advanced blue light blocking technology can protect your eyes, can effectively filter blue light, prevent glare, reduce eye fatigue, and help you fall asleep better.
Super clear and transparent, will not reduce the screen brightness, maintain the original screen brightness, and reduce eye fatigue.
The Protective Film adopts an oleophobic and waterproof coating, which can ensure anti-fingerprint, waterproof, dustproof and no air bubbles. Moreover, it is sensitive to touch, bringing natural touch, fast response and flawless precise touch.
If you want to know more about Imported Anti-Blue Light For ipad products, please click product details to view the parameters, models, pictures, prices and other information about Imported Anti-Blue Light For ipad products.
Whether you are a group or an individual, we will try our best to provide you with accurate and comprehensive information about Imported Anti-Blue Light For ipad!
Imported Anti-Blue Light Protective Film, Imported Anti-Blue Screen Protective Film, Imported Anti-Blue Screen Protector, Imported Anti-Blue Light Screen Protector
Shenzhen Jianjiantong Technology Co., Ltd. , https://www.tpuscreenprotector.com