Generally speaking, the ternary materials we often refer to are mainly NMC materials and NCA materials. The capacity of layered materials is affected by the structural stability. Since Ni3+ is more chemically stable than Co elements, The NMC material can also remove more Li during the charging process, so that the material capacity is greatly improved.
With the increasing energy density of lithium-ion batteries, traditional lithium cobalt oxide materials are gradually being replaced by higher-capacity ternary materials. Although ternary materials have a layered structure similar to LCO, they are compared to LCO materials. Ternary materials not only greatly improve the material capacity, but also have better thermal stability than LCO materials.
Generally speaking, the ternary materials we often refer to are mainly NMC materials and NCA materials. The capacity of layered materials is affected by the structural stability. Since Ni3+ is more chemically stable than Co elements, The NMC material can also remove more Li during the charging process, so that the material capacity is greatly improved.
Conversely, the structural stability of the layered oxide positive electrode material is also affected by the amount of de-separation. Excessive de-Li may cause the layered structure of the material to collapse. Therefore, in order to ensure the structural stability of the NMC material, the charge-off voltage of the material is required. Limitations are made to ensure long-term cycle stability of the material.
Johannes Kasnatscheew of the University of Münster, Germany, and their charging systems for NCM111 and NCM532 (both materials from the BMW Group), NCM622 and NCA (two materials from Customcell), and NCM811 (from Shanshan Technology) The effects of structural stability were studied.
Effect of charge cut-off voltage
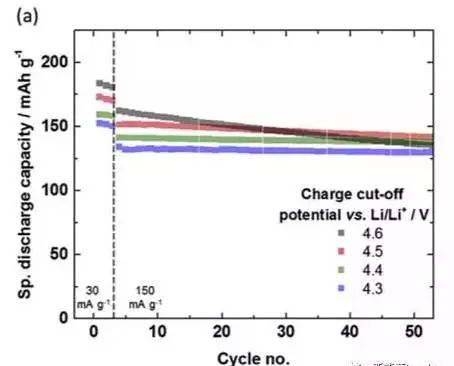
The amount of delithiation of the NMC material is proportional to the charge cut-off voltage, that is, the higher the charge cut-off voltage, the greater the amount of delithiation of the NMC material, and the more unstable the structure of the material. The figure below shows the cycle performance curve of the NCM811 material at different charge cut-off voltages. It can be seen that the material capacity is significantly improved after the cut-off voltage is increased, but the acceleration of the material decay rate is followed.
Comparing the cycle data under different cut-off voltages, it is found that the 4.6V cut-off voltage has the highest specific capacity at the fifth discharge, but after 53 cycles, its capacity drops rapidly, and the NMC111 is lower than 4.5V and 4.4V cut-off voltage. Capacity. This indicates that the charge cut-off voltage is improved blindly, although the capacity of the material is greatly improved, but the cycle stability of the material is significantly reduced, so it is necessary to select the charge cut-off voltage according to the design life of the battery. .
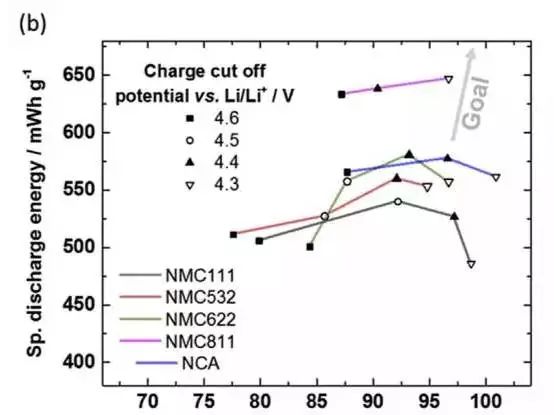
The following figure shows the discharge energy and discharge energy retention curves of NMC111, NMC532, NMC622, NMC811 and NCA materials after 53 cycles of different cut-off voltages. It can be seen from the figure that after 53 cycles, the discharge energy density The highest battery is not the highest cutoff voltage. For the NMC811 material, the highest discharge energy density is obtained at the 4.3V cutoff voltage. The NMC622 and NMC532, NCA materials have the highest discharge energy density at 4.4V charge cutoff voltage, and the NMC111 material is at 4.5. V obtained the highest energy density.
This is only the data after 53 cycles. As the number of cycles increases, the material at higher cutoff voltage is faster due to the decay rate. According to the trend of the cycle curve in the above figure, the discharge energy density will be the lowest when the cutoff voltage is lowest. Will be the highest. In addition, it can be seen from the following figure that no matter which material increases with the charge cut-off voltage, the capacity decay will be accelerated. In particular, the NMC111, NMC532 and NMC622 materials with lower Ni content are more affected by the cut-off voltage. This indicates that several materials with lower Ni content have less structural stability.
Environmental temperature effect
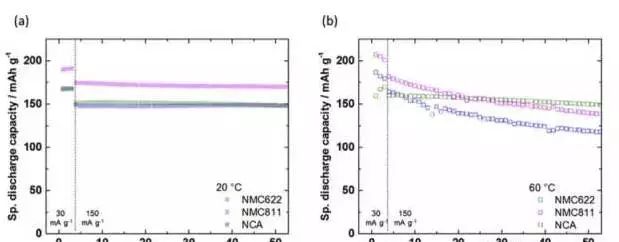
In the practical application of lithium-ion batteries, the high-temperature stability of materials is also considered. Johannes Kasnatscheew studied the cycling performance of NMC622, NMC811 and NCA materials at room temperature and 60 °C. The results are shown in the figure below. In general, increasing the temperature can improve the kinetic conditions in the battery, thereby improving the performance of the battery, which can be clearly seen from the capacity of the battery at 60 ° C, but the high temperature will have a certain degree of cyclic stability of the material. influences.
For example, at room temperature of 20 °C, the three materials have relatively close cycle performance in the first 50 cycles, but after increasing the temperature to 60 °C, the capacity retention rate of NMC811 and NCA materials after 50 cycles is significantly lower than that of NMC622. This indicates that the NMC622 material has higher thermal stability.
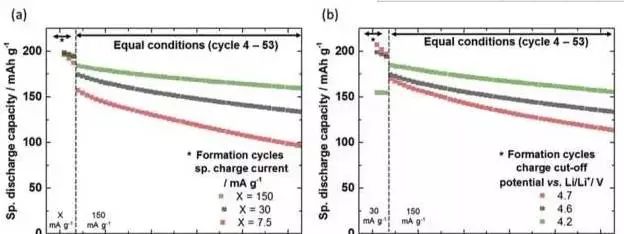
Effect of chemical current on cycle performance

Intelligent control of the charging system

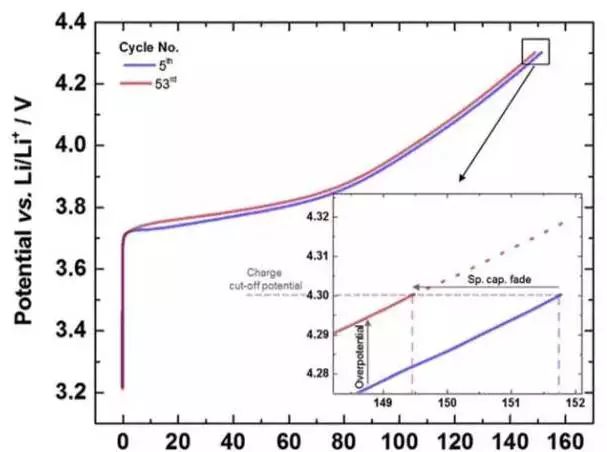
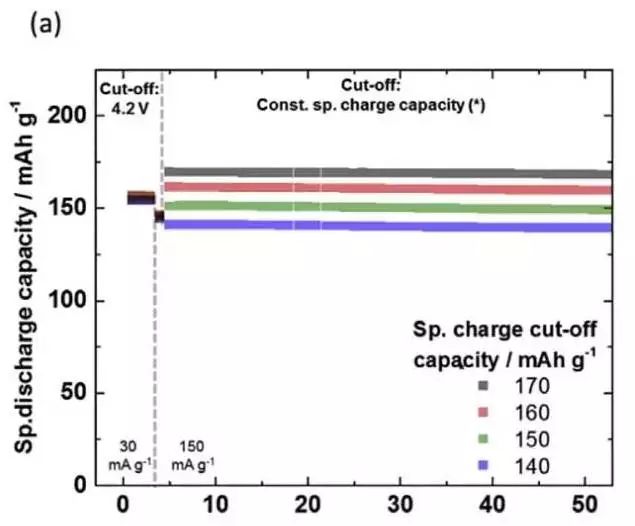
Johannes Kasnatscheew analyzed factors affecting the cycling performance of ternary materials, such as charge cut-off voltage and formed voltage and current, and the effect of ambient temperature on the cycle performance of NMC and NCA materials, essentially with the amount of lithium removed from the NMC material. The increase will lead to a decrease in the structural stability of the material and affect the cycle performance. In addition, high temperatures can have a negative impact on the stability of the material, resulting in a decrease in material cycle performance. Johannes Kasnatscheew also designed a new charging system based on the characteristics of NMC materials, which cuts off the charging voltage and ensures that the charging capacity of the battery is the same every time, thus overcoming the charging capacity due to the battery overpotential. And the decline of discharge capacity, a very good improvement of the cycle performance of the battery.
SHENZHEN CHONDEKUAI TECHNOLOGY CO.LTD , https://www.siheyidz.com