In recent years, the number of patients receiving bone marrow transplantation in the world has increased year by year, and the treatment trend of bone marrow transplantation has greatly improved the survival rate of patients. Bone marrow transplantation has been used clinically for many years, but it is still a high-risk treatment, mainly for patients with autoimmune diseases, blood cancers and rare genetic diseases.
Bone marrow transplantation, also known as hematopoietic stem cell transplantation, is a treatment method for the treatment of a series of diseases through intravenous transfusion of hematopoietic stem and progenitor cells to reconstruct the patient's normal hematopoietic and immune system.
Hematopoietic stem cell transplantation basically replaces the term "bone marrow transplantation" because hematopoietic stem cells are not only derived from bone marrow, but also from peripheral blood that can be mobilized by hematopoietic factors, and can also be derived from umbilical cord blood. These hematopoietic stem cells can be used To rebuild the blood and immune system.
Magenta Therapeutics (abbreviated as: Magenta) tries to develop safer and more effective stem cell transplantation technology, and is committed to solving the major needs of transplant medicine. The company mainly uses antibody-conjugated drugs, which can target and precisely eliminate the original stem cells, and use chemokines to mobilize stem cells more quickly and effectively. Magenta said that adding a small molecule called HSC835 to the culture medium can expand stem cells 400 times.
Through pioneering and innovative treatments, Magenta conducts in-depth research in four aspects: patient preparation before transplantation, stem cell expansion, stem cell collection, and post-transplant complications. Their goal is to maximize the application of bone marrow transplantation therapy to provide treatment assistance for patients with multiple sclerosis, scleroderma, acute myeloid leukemia, myelodysplastic syndrome, inherited metabolic diseases and sickle cell disease, and improve recovery effectiveness.
What has Magenta done so far?It is understood that Magenta is located in Cambridge, Massachusetts, and previously received a total of US$98.5 million in Series A and Series B financing. Magenta has attracted much attention in the Boston area with its lofty goal of improving stem cell transplantation.
In April 2018, Magenta received $52 million in Series C financing, led by Casdin Capita, and was oversubscribed by EcoR1 Capital, EvenTIde Asset Management, Watermill Asset Management and other long-term institutional investors. Mr. Eli Casdin, Chief Investment Officer of Casdin Capital, said that he believes that "the era of excellence in bone marrow transplantation" is coming.
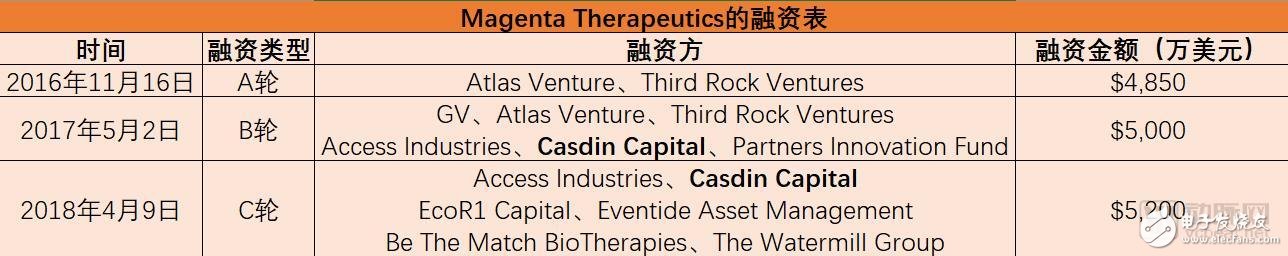
Magenta said that these funds will be used to promote the company's drug development portfolio, including targeted bone marrow transplant conditioning programs, stem cell mobilization and stem cell expansion plans.
In addition, Novartis (NovarTIs) highly recognizes the remarkable effect of Magenta in cord blood stem cell therapy, and has reached a mid-term drug licensing agreement with it. According to the agreement, Magenta has the right to use MGTA-456, a first-class allogeneic stem cell therapy.
In March 2018, Magenta and German biotechnology company Heidelberg Pharma jointly developed a bone marrow research and development agreement. The agreement stated that if Magenta exercises all target options and reaches its milestones, Heidelberg Pharma will receive up to 334 million U.S. dollars in revenue. This cooperation will combine Magenta's stem cell platform and proprietary antibodies, and achieve the goal through Heidelberg Pharma's antibody-targeted amanin binding (ATAC) platform.
Experience + academic = comprehensive understanding of stem cell transplantation
Dr. Jason Gardner, CEO, President and Co-founder of Magenta
"We are very grateful for the strong support of new and old investors, who share the same vision with us, so that more patients with debilitating diseases can benefit from bone marrow transplantation," Magenta's Chief Executive Officer (CEO), President and President Co-founder Dr. Jason Gardner said. "This financing provides us with additional funds, allowing us to continue to advance in clinical research and applications."
Dr. Jason Gardner joined Atlas Venture in November 2015. As a resident entrepreneur, he co-founded Magenta with Third Rock Ventures. Gardner has more than 20 years of experience in stem cell science and industry leadership roles.
Since 2005, Dr. Gardner has joined GSK and has been working for 11 years. He has served as the assistant director of clinical development, the vice chairman of the Center of Excellence for External Drug Discovery (CEEDD), the vice chairman of the Center for Regenerative Medicine, and the head of Boston Satellite R&D.
Dr. Gardner founded and led the Department of Regenerative Medicine, and established partnerships with the Harvard Stem Cell Institute and the Telethon Gene Therapy Institute. Their first stem cell drug, Strimvelis, was approved. Prior to this, Gardner, as the leader of CEEDD and a member of the clinical project team, collaborated to complete the late-stage development of Tykerb breast cancer and obtained the approval of the New Drug Application (NDA).
"This is the first time a company wants to understand stem cell transplantation so comprehensively," said Dr. Gardner. Their goal is to start a conversation about the risks/benefits of the therapy, so as to achieve technological changes and quality improvements. Magenta's mission is to focus more on the benefits of stem cell transplantation to patients, rather than amplify the risks it brings.
From 1995 to 1997, Dr. Gardner and Professor David Scadden of Harvard Medical School completed postdoctoral research on hematopoietic stem cells. Prior to this, he was educated in the UK and obtained a bachelor's and master's degree in natural sciences (biochemistry) from the University of Cambridge, and a doctorate in molecular medicine from the University of Oxford.
As the chairman of Magenta's board of directors, Mike Bonney is also the CEO of Kaleido Biosciences and has served as a partner of Third Rock Ventures. Kaleido BioSciences is an American biomedical technology company dedicated to discovering and developing new chemicals to drive the organ function of the microbiome. The company is currently independently researching and developing microbial metabolism therapies, covering rare genetic diseases, metabolic diseases, oncology and other diseases.
From 2003 to 2014, Bonney served as the CEO and director of Cubist PharmaceuTIcals. Under his leadership, Cubist PharmaceuTIcals has grown from a struggling micro-company to the world's leading antibiotic company. In early 2015, the company was acquired by Merck & Co for $9.5 billion.
In addition, as a native of Boston, Bonney graduated from Bates College before entering the pharmaceutical industry. He was hired by Biogen in 1995 and eventually became the company's vice president of sales and marketing.
During his tenure at the company, Bonney developed Avonex. Avonex is a beta interferon drug that can delay multiple sclerosis and reduce the chance of clinical deterioration. MarketWatch, a New York-based financial information website, claims that this is one of the most successful drugs in the history of biotechnology.
The four stages of stem cell transplantation and drug development
"Hematopoietic stem cells have a remarkable ability to generate new cells in our blood and immune system. Over one million patients have undergone cell transplantation in the past 50 years," said Dr. Gardner, CEO of Magenta. "Technical and scientific limitations have made stem cell transplantation the last resort to treat deadly diseases, but new science is ready to enter the clinic and can fundamentally open up new drugs for patients suffering from early-stage cancers, blood diseases and autoimmune diseases, including Multiple sclerosis and scleroderma, etc."
Magenta said that their new targeted drug treatments can reduce complications after transplantation, such as graft-versus-host disease (GVHD).
GVHD is due to the T lymphocytes in allogeneic donor transplants after transplantation, which are stimulated by a series of "cytokine storms" initiated by the recipient, which greatly enhances its immune response to the recipient's antigen, and starts the cells with the recipient's target cells as the target. Poisonous attack, of which the skin, liver and intestines are the main targets. The incidence of acute GVHD is 30%-45%, and the incidence of chronic GVHD is lower than that of acute.
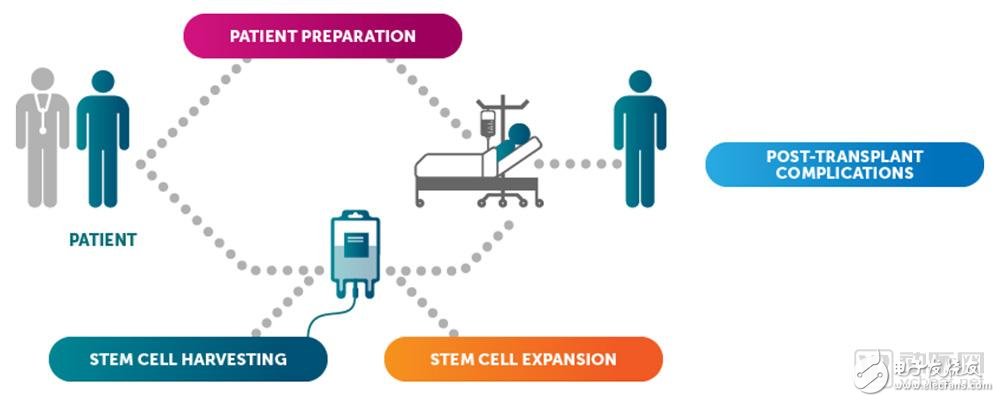
Scientists at Magenta have proven that using related drugs, they can recognize stem cell antibodies and selectively remove stem cells and diseased cells under safe conditions.
Magenta said that improved targeted conditioning agents can extend the use of bone marrow transplantation therapy to patients who cannot use the therapy, as well as patients who currently believe that bone marrow transplantation is too dangerous.
In the pre-transplant preparation phase, Magenta uses C100 (attenuation of hematopoietic stem cells and immune cells) and C200 (attenuation of hematopoietic stem cells) and C300 (attenuation of immune cells) for pre-clinical drugs.
Stem cell expansionMagenta is committed to developing new methods that can not only expand stem cells, but also maintain the capabilities required for cell transplantation. These methods can deliver the optimal number of healthy stem cells to patients, greatly improving the results after stem cell-based therapy.
Magenta uses the most advanced clinical stage plan MGTA-456 to perform inspections for various transplant environments. By expanding the number of umbilical cord blood stem cells, MGTA-456 has enough potential to improve the survival rate of patients and enable more patients to obtain high-quality cord blood units matched by HLA.
MGTA-456 is a cord blood expansion product obtained by culturing cord blood stem cells on a medium containing aryl hydrocarbon receptor antagonist, SCF, Flt-3L, IL-6 and TPO for 15 days. In April 2018, MGTA-456 obtained orphan drug designation from the U.S. Food and Drug Administration (FDA) and was designated for patients undergoing bone marrow transplantation.
MGTA-456 has obtained clinical evidence to prove its concept in phase 1 and phase 2 clinical trials of 36 patients with blood cancer. Clinical trial data show that 36 patients who received MGTA-456 treatment have all been successfully transplanted, and meanwhile, the average recovery time of transplantation has been accelerated.
In the stem cell expansion stage, Magenta uses MGTA-456 (to expand umbilical cord blood stem cells in vitro) for clinical treatment, and MGTA-E200 (to expand in vitro stem cells for gene therapy/editing) is used in pre-clinical phase.
Stem cell collectionThe new model developed by Magenta can stably mobilize a large number of stem cells into the blood within a day, thereby providing more patients with a high-quality source of stem cells. Stem cells have the potential to proliferate and differentiate, have the ability to self-renew and replicate, and can produce highly differentiated functional cells.
In the stem cell collection phase, Magenta's pre-clinical drugs are MGTA-M100 (mobilization of hematopoietic stem cells).
Complications after transplantationMagenta is seeking new therapies that can target GVHD T cell donors and reduce the occurrence of acute GVHD without reducing the beneficial effects of the donor graft.
According to the arterial network, GVHD caused by the immune response mediated by donor T cells is an important factor affecting the quality of life of patients after transplantation, and it is also the main cause of complications and death after transplantation.
In the stage of post-transplant complications, Magenta is still discovering the drug G100 (attenuation of the immune response activated by the allogeneic).
The difference between bone marrow transplantation and peripheral blood stem cell transplantationThe purpose of both is to collect and transplant hematopoietic stem cells, plant them into patients, and maintain their hematopoietic and immune functions. However, bone marrow transplantation requires about 750ml-1000ml of bone marrow to be collected under anesthesia, which brings certain pain and inconvenience to the donor.
Since it was discovered that hematopoietic stem cells can be mobilized into the blood and collected by a blood cell separator, most of the current transplants use peripheral blood stem cell transplantation. Compared with bone marrow transplantation, it has the advantages of faster hematopoietic recovery, convenient collection, less pain for the donor, easy acceptance by the donor, and no impact on the health of the donor.
High definition LED displays or HD led screen also called Small pixel pitch led display screens represents the latest and
top technology of LED display industrial, they are widely used in high-end
conference
rooms, TV studios, the government mansion, etc. With 400mm*300mm cabinet
size, it is very easy to get 16:9 or 4:3 display ratio. As the 400*300mm cabinet is light weight and has high precision in cabinet size.
HD LED Video Wall ,HD LED Video Wall, indoor LED Display, Indoor LED screen
Shenzhen Priva Tech Co., Ltd. , https://www.privaled.com